
IMRAM
Institute of Multidisciplinary Research for Advanced Materials, Tohoku University
東北大学
多元物質科学研究所

LAST UPDATE 2021/05/01
-
研究者氏名
Researcher Name南後恵理子 Eriko NANGO
教授 Professor -
所属
Affiliation東北大学 多元物質科学研究所
有機・生命科学研究部門
Institute of Multidisciplinary Research for Advanced Materials, Tohoku University
Division of Organic- and Bio-materials Research -
研究キーワード
Research KeywordsX線自由電子レーザー
結晶構造解析
時分割測定
X-ray free electron lasers
Crystallography
Time-resolved measurement
- 研究テーマ
Research Subject -
タンパク質ダイナミクス解析と分子制御への応用
Analysis of protein structural dynamics and its application to molecular control
研究の背景 Background of the Research
細胞情報伝達、生体内触媒反応、貯蔵や輸送を行うなど、生命現象を支える重要な生体構成物質であるタンパク質は、多数のアミノ酸から構成され、その立体構造は機能と深く関連していることが知られています。今までに16万を超える多くのタンパク質構造が決定されてきましたが、そのほとんどが“止まった”状態の構造であり、機能が発現される動的過程の原子レベルでの解明が期待されています。
A protein is one of the biomolecules that are essential to the phenomena of life, such as cell signaling, in vivo catalytic reactions, storage and transport. A three-dimensional structure of a protein is closely related to its function. While many protein structures over 160,000 have been determined, most of them are “static structures” under cryogenic temperature. Therefore, it is desired that the dynamic process in which proteins function is elucidated at the atomic level.
研究の目標 Research Objective
X線自由電子レーザーや放射光などの量子ビームを用いて、タンパク質の中で実際に起こっている化学反応や構造変化を可視化する技術開発を行い、光に応答するタンパク質のスイッチ機構や、ユニークな反応を触媒する酵素の反応機構などの解明に取り組んでいます。また、得られた精密な構造情報を基にタンパク質分子の合理的設計と新機能をもつ分子の創製を目指します。
We are developing techniques using a quantum beam such as an X-ray free-electron laser or synchrotron radiation to visualize actual chemical reaction and structural changes in proteins. We will reveal the reaction mechanism of light-sensitive proteins and the reaction mechanism of enzymes catalyzing unique reactions. Furthermore, we aim to design and create protein molecules with new functions based on precise information from dynamic structural analysis.
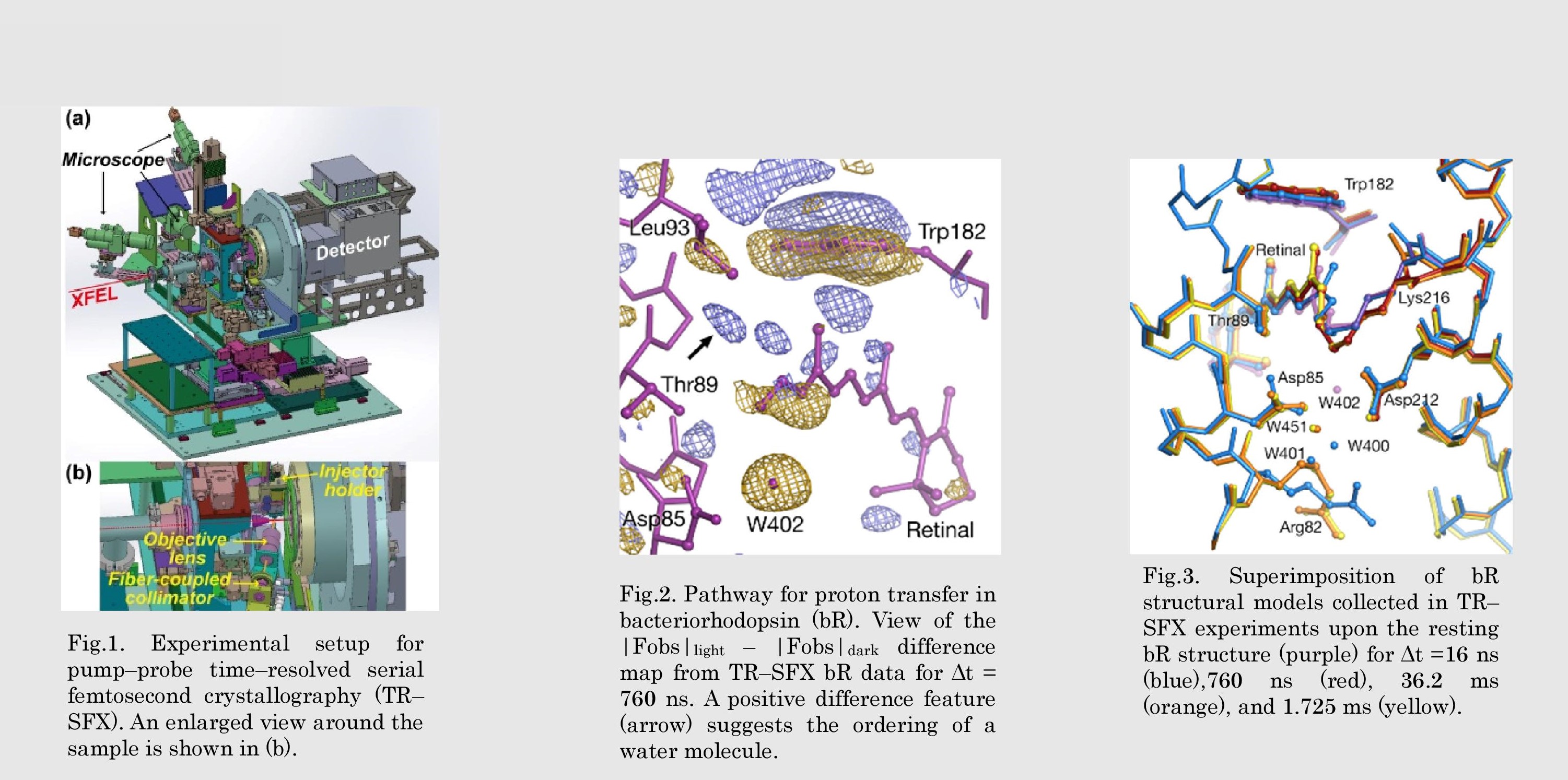
研究図Figures
論文発表 / Publications
PNAS 119, e2117433119 (2022). Struct. Dyn. 7, 024701 (2020). J. Appl. Cryst. 52, 1280 (2019). Science 261, 145 (2018). Nat Commun. 8, 1585 (2017). J. Synchrotron Radiat. 24, 1086 (2017). Nature 543, 131 (2017). Science 354, 1552 (2016).
研究者連絡先 / HP
- eriko.nango.c4
 tohoku.ac.jp
tohoku.ac.jp - http://www2.tagen.tohoku.ac.jp/lab/nango/html