
IMCE
Institute for Materials Chemistry and Engineering, Kyushu University
九州大学
先導物質化学研究所

LAST UPDATE 2017/02/25
-
研究者氏名
Researcher Name友岡克彦 Katsuhiko TOMOOKA
教授 Professor -
所属
Affiliation九州大学 先導物質化学研究所
分子集積化学部門・集積分子機能分野
Institute for Materials Chemistry and Engineering, Kyushu University
System of Functional Molecules, Division of Applied Molecular Chemistry -
研究キーワード
Research Keywords有機合成化学
キラル分子化学
不斉合成化学
構造有機化学
organic synthesis
chiral molecule chemistry
asymmetric synthesis
structural organic chemistry
- 研究テーマ
Research Subject -
非天然型キラル分子の創製と応用
Synthesis and Application of Unnatural Chiral Molecules
研究の背景 Background of the Research
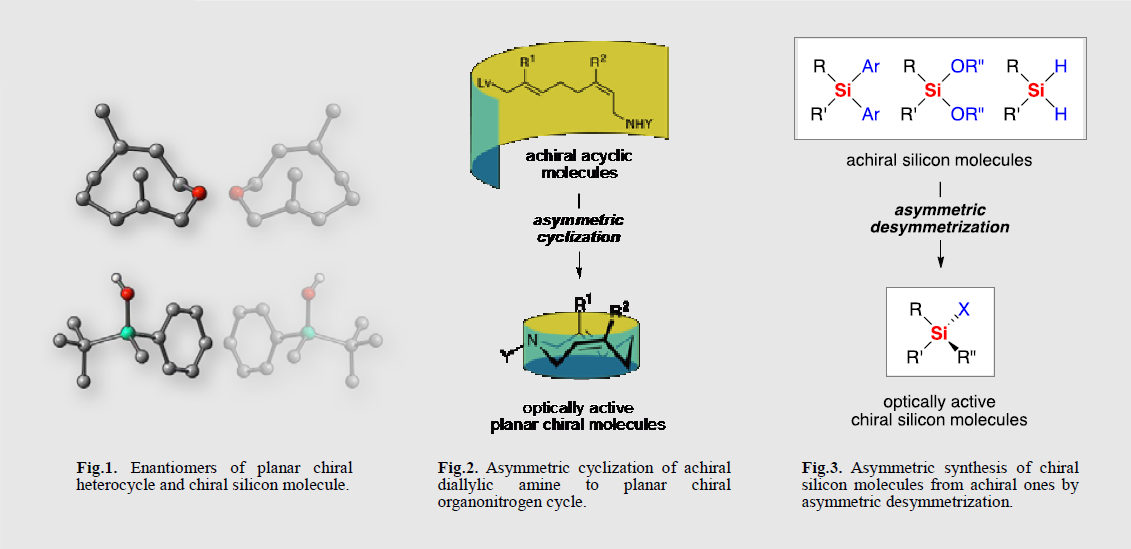
キラル分子の化学に関してはこれまで膨大な研究が行われてきたが,その多くが天然に豊富に存在する炭素中心性不斉を有する分子を対象とするものであった.これに対して我々は,天然に存在しないキラル分子(非天然型キラル分子)の創製とその基礎および応用研究の展開を目指して,主として面不斉を有するヘテロ中員環分子とケイ素中心性不斉を有するキラルケイ素分子について研究を行なっている.
Most of naturally occurring chiral molecules comprise asymmetric carbon, hence our effort on asymmetric synthesis mainly concenter a stereocontrol of central chirality of carbon. On the other hand, it is also known that molecular chirality can be caused by a presence of central chirality of non-carbon element or by a restricted rotation about single bonds. Among those unnatural chiral molecules having no asymmetric carbon, we have particularly studied on planar chiral heterocycles and chiral silicon molecules.
研究の目標 Research Objective
我々の研究目標は,1)面不斉ヘテロ中員環分子の特異な動的立体化学挙動や高度に規制された立体配座を活用した光機能性分子や不斉反応剤の開発,2)キラルケイ素分子の特性を活用した新しい生体機能分子の開発である.そのために,これらの非天然型キラル分子の汎用的な不斉合成法を開拓するとともに,基礎的な物性や反応性について精査している.
Our research objectives are, 1) development of planar chiral heterocycle-based optical functional molecules and asymmetric reagents having unique functionalities caused by the dynamic chirality with highly restricted conformation, 2) development of chiral silicon-based bio-functional molecules having unique functionalities caused by the steric and electronic effects of the silicon. To this end, we have been studied on practical asymmetric synthetic methods for the unnatural chiral molecules along with the researches on physical properties and reactivity thereof.
研究図Figures
論文発表 / Publications
J. Am. Chem. Soc., 127, 12182 (2005). Org. Lett., 8, 963 (2006). Tetrahedron Lett., 49, 6327 (2008). J. Am. Chem. Soc., 132, 9232 (2010). J. Am. Chem. Soc., 133, 1754 (2011).
Angew. Chem. Int. Ed., 51, 10355 (2012). J. Chem. Eur. J., 20, 7598 (2014). Angew. Chem. Int. Ed., 54, 1190 (2015).
研究者連絡先 / HP
- ktomooka
 cm.kyushu-u.ac.jp
cm.kyushu-u.ac.jp - http://www.cm.kyushu-u.ac.jp/tomooka/tomooka